Coolant Considerations For Medical Machining
While the proprietary nature of the actual metalworking processes used for making 'body parts' tends to be more mystique than technique, there are some things to think about for shops that are looking to do this kind of work.
It’s no secret that one of the very few bright spots in the metalworking business has been the machining of medical components. It only makes sense if one considers the demographic glut of the aging baby boomer generation.
More bodies are reaching a point where replacement parts are needed. Demand creates supply with so many of us among that aging human fleet.
Like much of the high tech label that surrounds medicine and its practice, some shops are hesitant to consider medical machining applications because they believe the special demands may be beyond their capability. There are exceptions, but in general the metalworking process knowledge to make medical components is generic. Cutting metal is cutting metal, and although some of the materials specified for medical components might be considered “exotic” they are certainly not exclusive to medical applications.
One area of medical machining where a shop looking to create a successful process might need to understand a few things is the application of metalworking fluids. In general, traditional machining guidelines for fluid use are in effect for most medical machining applications, but there are some considerations in applying them that can help make the job easier.
We spoke to Tom McClure, manager of the fluids business at TechSolve (Cincinnati, Ohio), and Greg Foltz, engineering and development manager for Cimcool at Milacron Inc. (Cincinnati, Ohio), to learn more about the role of metalworking fluids in machining of medical components.
Medical Parts
There are two main categories of machined parts for medical applications. They are components that are implanted in the body on a permanent or semi-permanent basis and those that are used in the body for short durations, such as during surgery. Most of the specialized cutting fluid considerations are targeted at the transplanted parts.
Implanted components such as hip joints, knee joints, bone screws, spinal fusion plates and other hardware must be designed to withstand a relatively tough environment within the human body. Many of these components are specified to be made from tough materials such as titanium and nickel alloys to withstand a hopefully long service life within the acidic environment that exists in our bodies, which have a general pH of around 5.4.
Greatly simplified, on skeletal replacement or reinforcement parts, the hope is that once the component is in place, the surrounding tissue will adhere to the surface of the metal—literally grow on it to form a strong connection—like a weld. This critical union between body and part is where what the shop did upstream through its application of metalworking fluid and the subsequent cleaning of the parts can be a determining factor in the transplant’s success.
Oil Is OK, But Be Careful
According to Mr. McClure, most of the medical machining shops that TechSolve has worked with use water-based coolants in their medical parts manufacturing. “That’s not to say oils cannot be used,” he says, “but the cleaning of those components must be especially thorough.”
It seems there was a lawsuit filed against a medical parts supplier after a batch of implanted parts failed to knit with the surrounding tissue. These had to be removed and replaced. No doubt the malpractice lawyers had a field day.
Analysis showed that microscopic amounts of the cutting oil were present in the microstructure of the implanted part. This residue was sufficient to prevent the tissue from bonding to the component. This is a patient’s horror story for sure, and a cautionary tale to parts suppliers.
Obviously if cutting oil is the right fluid for the job, the shop should use it. But it is important to make sure the subsequent cleaning process can get the oil off the parts.
Coolant Considerations
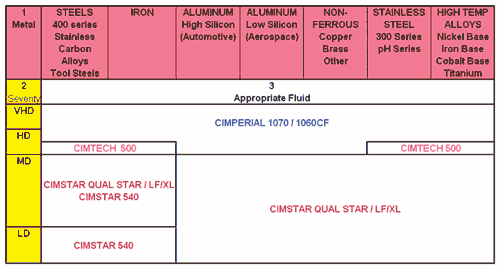
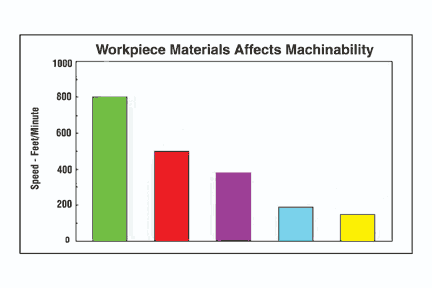
For medical component applications, coolant selection is a twofold exercise. First, of course, are the performance characteristics created by the interaction among the coolant, cutting tool material and the workpiece material. These are generic concerns applicable to any metalworking process.
Looking then at synthetic and semi-synthetic coolants, what should the shop think about? Matching the right coolant to the job is, of course, key. It is also important to reduce any incidences for bacterial growth in the coolant. According to Mr. McClure, most shops doing medical machining make coolant cleanliness a higher priority than is generally seen in traditional shops.
Coolant tanks are covered, and nothing foreign is allowed into the system. Way oil is monitored and removed regularly. Many shops use individual coolant tanks, one for each machine. In the case of an outbreak of bacteria or other contamination, dumping a single tank is less disruptive to production than dumping a central system, and it isolates any problems to a single machine instead of the entire shop.
Flood coolant application is generally the application system of choice for implanted medical parts cutting. High pressure coolant systems are not widely used for fear they can deposit coolant in pores of the metal, complicating cleaning.
Clean parts are the bottom line consideration for machining implanted medical components. Coolant selection plays a role in this.
Complicating the making of implanted parts is the capability to remove any residue from the parts that is used in their manufacture. In other words, if you can’t clean it, don’t use it.
As an example, many shops rely on ultrasonic parts cleaning for complex, cavity riddled workpieces. Such cleaning systems work well at getting into nooks and crannies on a workpiece. However, some workpiece materials are sensitive to ultrasonic, which can induce micro-fractures in the workpiece.
Think Aerospace
For shops looking at machining medical parts, both Mr. McClure and Mr. Foltz agree that the best model to follow is machining of aerospace components, especially engine parts. If you think about it, it’s true. Aerospace manufacture is regulated by the FAA, which imposes traceability and process qualification standards for making the parts that go into commercial and military aviation.
The medical parts business is under the auspices of the FDA, which likewise demands that parts be traceable to the source and qualifies production processes as meeting the regulatory standards. In both industries, once a process is qualified, there is some inertia to make changes because of the regulatory red tape involved. Getting the process correct and sustainable should be a goal for machining medical components.
Related Content
Arch Cutting Tools Acquires Custom Carbide Cutter Inc.
The acquisition adds Custom Carbide Cutter’s experience with specialty carbide micro tools and high-performance burrs to Arch Cutting Tool’s portfolio.
Read MoreANCA Introduces Tool Loading System for Medical Drills
ANCA’s dental drill loading kit includes tailored RoboMate pallets for FX machines using a FANUC 200iD robot loader.
Read MoreBeyond the Machines: How Quality Control Software Is Automating Measurement & Inspection
A high-precision shop producing medical and aerospace parts was about to lose its quality management system. When it found a replacement, it also found a partner that helped the shop bring a new level of automation to its inspection process.
Read MoreEDM, Laser Micromachining and More at GF Medical Demo Center
At GF’s Medical Center of Competence, the company shows off EDM and laser features that could make a large impact on medical manufacturing — and elsewhere.
Read MoreRead Next
The Cut Scene: The Finer Details of Large-Format Machining
Small details and features can have an outsized impact on large parts, such as Barbco’s collapsible utility drill head.
Read More3 Mistakes That Cause CNC Programs to Fail
Despite enhancements to manufacturing technology, there are still issues today that can cause programs to fail. These failures can cause lost time, scrapped parts, damaged machines and even injured operators.
Read More












.png;maxWidth=300;quality=90)





.png;maxWidth=300;quality=90)











