Accessing Hidden Capacity in Medical Device Manufacturing
(MMS Online Exclusive) Data collection is an essential step in process improvement, allowing engineers to determine root causes and to ensure processes remained fixed permanently. The CNC machine tool is capable of capturing and communicating the status of operations and can be incorporated into a comprehensive automated data collection and analysis infrastructure that allows assigned resources to focus on implementing the best process improvements.
Introduction
The medical device market continues to experience double digit growth. The orthopedic device and implant market exceeds 10 billion dollars and is forecasted to grow to 18 billion dollars by the year 2009. When companies grow they tend to ignore cost controls and focus on getting the additional capacity required to fill customer demands before a competitor provides an alternative source of supply. Medical device manufacturers have not been motivated to focus on costs in the past because compliance and quality have been more important, and the industry has had the luxury of operating with a cost-plus pricing model.
The industry is now facing new levels of price pressure as its customers try to control their soaring health costs. This is compounded by the fact that medical devices are often manufactured in low volumes, which often leads to inefficient machining processes.
It is not surprising then that medical device manufacturers are looking to Lean Manufacturing and other continuous improvement methodologies to improve processes, reduce costs and maximize the capacity of existing assets. Lean incorporates the concepts of Total Productive Maintenance that utilizes the Overall Equipment Effectiveness (OEE) metric to drive continuous improvements in process availability, performance and quality.
Data collection is an essential step in process improvement, allowing engineers to determine root causes and ensuring processes remain fixed permanently. The CNC machine tool is capable of capturing and communicating the status of operations and can be incorporated into a comprehensive automated data collection and analysis infrastructure that allows assigned resources to focus on implementing the best process improvements.
Unique Challenges
Traditionally, sustained growth has meant that manufacturing is required to expand capacity by investing in new plant equipment. The increased revenue alone typically justifies the investment, and manufacturing either replicates existing processes or tries to introduce new and more efficient processes.
However, for medical device manufacturers, simple expansion may not be an option due to other manufacturing challenges:
- Compliance – adding equipment is only the first step; new workers must be found and trained to work in a highly controlled work environment.
- Quality – likewise, stringent quality standards must be maintained as new workers, equipment and processes are brought on line.
- Increased competition – global competitors may be ready to offer capacity immediately and at a reduced cost.
- Price pressures – global competitors and domestic group buying practices limit the actual revenue growth potential, despite the need for more product volume.
- Low-volume, high-mix – more choice means that existing manufacturers need to offer more product variations and appeal to different market segments.
Adding capacity in medical device manufacturing has to be achieved with more care than may be required in many other industries. For the same reasons, it is very difficult to solve the problem quickly through increased outsourcing; therefore many manufacturers are focusing more earnestly on existing asset utilization.
In Search of the Perfect Process
Lean, Six Sigma, Total Productive Maintenance, Theory of Constraints, the Toyota Production System and a host of other popular manufacturing toolkits all “share the goal of searching for the perfect process” through continuous improvement. They only differ in their starting point and emphasis.
The perfect process is one in which every step has the following characteristics.
- It is valuable, meaning there is no waste. That is, it includes nothing that the customer isn’t willing to pay for.
- It is capable, so that it gives good results every time, from the first setup part to the last production part.
- It is available, so that it produces when needed. That means that the machine, the tooling, the materials and labor are all available at the right time.
- It is adequate, meaning that it can produce at the required production rate with no bottlenecks.
- And finally it is flexible, meaning that it can accommodate quick changeovers from one product to the next.
With the exceptional challenges in growth, compliance, quality and increased competition facing the medical device industry, it is not surprising that many manufacturers are emphasizing perfecting their existing processes.
Data Driven Continuous Improvement
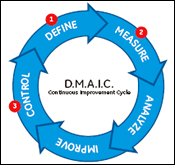
Many continuous improvement programs use variations of either the Demming Cycle (Plan-Do-Check-Act) or the Six Sigma D.M.A.I.C. Cycle (Define-Measure-Analyze-Improve-Control). Both cycles use data to determine root causes and to ensure that any improvements made are effective. Data is collected three times in the D.M.A.I.C. cycle (Figure 1):
- In the define stage, identifying which critical to quality (CTQ) process issues effect the customer.
- In the measure stage, determining the values of process variables that impact the CTQ.
- In the control stage, to confirm that the improvements made have been effective and that they have positively impacted the CTQ.
Without data, it can be tempting to assume that current processes are similar to other situations that have been experienced, and facts may be twisted to fit these preconceptions.
Data collection helps us to determine which investments will be most effective. Without credible data, it is becoming increasingly difficult to convince senior management to invest the time and materials to achieve the necessary improvements. Without data and analysis to discover the root causes of problems, improvements may only work temporarily or for special cases.
It is challenging to decide what data needs to be collected and how the data needs to be grouped and analyzed to produce meaning. Lean and Total Productive Maintenance consider the three primary metrics of availability, performance and quality, and combine them into a comprehensive Overall Equipment Effectiveness (OEE) metric.
Consider an OEE measurement for a single shift of production. An operation plans a 30 minute break and two 15 minute breaks. There was 47 minutes of downtime. The machine is capable of producing 80 parts per hour, and 413 parts were produced in total, 23 of which were not produced to specification.
Availability is calculated by dividing the Actual Operating Time by the Planned Production Time. This metric focuses on setup and downtime issues.
Performance is calculated by dividing the actual parts produced by the number of parts that should have been produced at the ideal rate. This metric focuses on the speed of the process and the impact of any minor stoppages. The Quality metric is calculated by dividing the number of good parts produced by the total number of parts produced. This metric evaluates scrap rates during setup and production. The three components are individually calculated, then multiplied together to come up with the OEE rate.
Most companies do not know how efficiently their machining operations perform. Even if they track the status, they have insufficient insight into the root causes that drive the metrics. World-class organizations in a variety of industries are achieving OEE metrics of over 80%. Without focus, many machining operations may be achieving less than 40%.
Challenges with Machine Tool Efficiency
The low-volume, high-mix machining environment that is typical in medical device manufacturing is prone to inefficiency.
Machine tool setup is a sequence of time consuming measurements and data entry steps to establish tool geometry, work piece location and tool wear offsets in the CNC. Aside from being time consuming, these measurements and the data entry procedures are sources for variation and error.
There are several sources of downtime on a machine tool, most of which are related to machine crashes. Crashes can occur for a variety of reasons. Perhaps standard procedures were not followed? Perhaps the fixture, the blank or the part program did not match? Mechanical failures often turn up after a crash, but failures can also be attributed to a lack of regular maintenance.
There are also several reasons why a machine may be running slow. Some CAM system post processors are notoriously inefficient, and feeds and speeds are often conservative. Machines are sometimes programmed to run slow because the machine cannot follow a complex profile accurately at high speed, or there are work holding limitations. Process variations such as material hardness, blank size, tool wear, and variations in cutting width and depth may force a worse-case feed rate to be programmed. There are also several sources of minor stoppages that affect the efficiency of the machine tool. Tool wear is a basic issue and tools may be replaced prematurely, either due to inappropriate feeds and speeds, or to try and avoid a potential breakage. High-value parts may have to be checked, and offsets updated, before the final pass to ensure critical dimensions. Large parts may have to be inspected on the machine, because it is difficult to reestablish the part reference points if the part is unloaded. A machine may sit idle waiting for attention, while the operator services another machine.
Setup part yield is very important in short run production. Given the high cost and the shortages of the materials commonly used in medical device manufacturing, it would be advantageous to achieve 100% yield. Machine accuracy may limit setup yield because even a simple 3-axis machine tool has 27 degrees of freedom, and a problem in any of these can cause errors in the parts produced. Setup errors may also scrap parts.
Production yield tests a machine’s repeatability. Unless a machine tool is in very bad condition, it should be able to repeat the specification established during setup, subject to all the other ongoing manual processes being executed flawlessly.
A machine tool operation may face all of these challenges in varying degrees, and there are several robust solutions to each of the problems discussed. The typical process improvement solution priorities of eliminate, automate or error-proof apply to these problems. Since there are limited funds it makes sense to apply investments to those areas with the greatest potential for improvement for a specific machining operation. This will improve the process capabilities most effectively and drive the OEE metrics.
Manual Data Collection
Some manufacturers initially try to collect data manually, but they are often disappointed by the quality and impact of the data collected.
Without adequate employee training and motivation, there can be significant differences with how individuals collect data, skewing results. Poor forms or inadequate training can lead to data entry errors. The process of data collection can change behaviors in such a way that the “typical process” is not captured. Operators may slow down or speed up, depending on their motivation. Unless the data collection is a consistent part of their process, they may forget to enter the data, and they may even fabricate or manipulate data to cover up issues they find threatening.
The resources consumed by recording, collecting, data entering and analyzing manual data may mean there are no resources left to actually improve the process. It may be impossible to report results in a timely manner so that actions can be taken to correct problems before they impact the business.
Manual data collection can be enhanced with low-cost, electronic data entry terminals located close to the process. Electronic data entry speeds up the process of getting the data into an electronic form for analysis. Just like paper forms, it is important to optimize the data entry screens for simplification and understanding, and to minimize keystrokes. Electronic displays also provide a medium to display status and trend information back to the operators. Providing timely feedback is an essential part of achieving buy-in from the whole team. Manual data collection will always be resource intensive, and subject to errors and bias. Bad data will produce bad analysis, and any process improvements will be less effective.
Automatic Data Collection
Powerful, intelligent industrial controls such as CNCs, robot controllers and PLCs are at the center of most modern machining operations, and they include Ethernet connectivity that provides access to a wide range of information and process events. Older controls can also be connected to an Ethernet data collection system via a variety of smart communication conversion modules.
For example, the CNC knows what parts are being produced and can count them. It knows the general status of the machine tool and any auxiliary equipment because of extensive diagnostics built into the CNC and the machine tool builder. It knows about operational modes, so it can typically distinguish between production and setup. It can report on feed rates and any overrides initiated by the operator. And with integrated gauging or with feedback from an external gage or coordinate measurement machine, it can determine which parts are produced to specification and which ones are bad.
If everything looks operational to the CNC, but parts are not being produced, it can ask the operator to enter a reason for the wasted production opportunity. This will capture exceptional process issues such as no material, meeting attendance, and unplanned maintenance.
Electronic data collection not only overcomes the errors and bias of manual data collection, but it can also fully automate the data entry, organization, and much of the analysis of the data. Information can be shared across business functions so corrective actions can take place in real-time.
There are additional benefits provided by the connectivity installed for data collection:
- High-speed data transfer of part programs, subprograms, macros, parameters, work offsets, tool offsets and in-cycle measurements.
- Remote diagnostics means that maintenance staff can often diagnose or even recover from a problem without visiting the machine, or at least turn up to the machine with parts based on a preliminary diagnosis.
- Remote backup of critical system files to minimize downtime in the event of a system memory failure.
Automated data collection and reporting can be implemented at a machine, cell, factory and the enterprise level. The efficiency data can be correlated with other systems such as ERP and quality systems to see if factors interact. Information can be integrated with business systems and computer maintenance management systems (CMMS). Automated data collection can be an important part of a compliance assurance program.
Turning Data into Information
Once the data is captured electronically, reports can be automatically generated in real time to satisfy each stakeholder, including production, maintenance, quality control and business functions.
For most people, raw data is not very informative. The famous statistician Ellis Ott always encouraged his students to plot the data, because the picture provides new insight.
Visual analytics turn raw data into actionable information. Several levels of data analysis are often required to satisfy a broad range of stakeholders.
High-level, dashboard screens with easy navigation and intuitive drill-down capabilities will typically satisfy those stakeholders that are only interested in status. More comprehensive reports with details that allow comparative analysis will be required for those responsible for actually improving processes. Electronic real-time information portals can combine data with information from external sources to evaluate correlations. Low-level access to some of the data may be required for more complex analysis using common analytical tools such as Microsoft Excel or specialized statistical tools such as Minitab.
The human brain is very fast at identifying patterns in visual data. Manufacturing with automation should appear very rhythmic over time. Materials are loaded, the machine cycles and the finished part is unloaded. Displaying production events and duration graphically makes any anomalies stand out as they break the expected repetitive patterns.
One of the most powerful data analysis strategies is to use comparative data, looking for best practices to adopt and bad practices to eliminate through process change or training. For example, do certain products or part numbers create more or less downtime then others? Are there differences between crews or shifts? What are the mean-time between failure and mean time to repair statistics?
Drill-down analysis may provide a deeper understanding of the root causes. Do certain problems occur more often than others? Are they special cause problems that only happen occasionally, or are they systemic problems that reoccur day after day, or week after week. Obviously, focusing on problems that impact production capacity the most will prioritize process improvement actions.
Grouping problems can also provide insight. Are the key problems related to machinery, processes, people or some other category? Do certain processes or departments have more issues than others? This type of analysis can help select future projects for more in-depth study.
Summary
Most companies understand that continuous improvement is an essential part of their competitive strategy. Medical device manufacturers need to generate more capacity with their existing assets, and reduce costs to respond to price pressures from the market and the competition.
New machine commissioning problems and lengthy new operator learning curves are also avoided by leveraging the hidden capacity in existing assets, often solving quality and compliance issues.
Data collection is necessary and a major challenge for continuous improvement projects. Manual data collection is subject to errors and bias. It can also use up all the resources available for process improvement. For machining operations, the CNC is at the center of most processes, and it includes the technology to allow automatic data collection that can characterize the process and uncover root causes. Analytical tools can provide important insight, especially when a variety of comparative views are available.
The outlook for medical device manufacturing is strong for the foreseeable future, but it is critical for the existing manufacturers and suppliers to respond to the need for growth and cost control. Action must be taken to satisfy all the stakeholders including customers, investors and employees, and to sustain the profit margins that fund future product development.
Read Next
3 Mistakes That Cause CNC Programs to Fail
Despite enhancements to manufacturing technology, there are still issues today that can cause programs to fail. These failures can cause lost time, scrapped parts, damaged machines and even injured operators.
Read MoreThe Cut Scene: The Finer Details of Large-Format Machining
Small details and features can have an outsized impact on large parts, such as Barbco’s collapsible utility drill head.
Read More










.png;maxWidth=300;quality=90)














